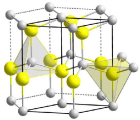
3. What are the properties of ZnO nanoparticles?
Information has been provided on nano-sized ZnO raw materials (source materials as powder for sunscreen formulations) and some final cosmetic products containing some of the nano-sized ZnO raw materials. Information has also been provided on selected raw materials from various manufacturers, in the form of both coated and uncoated nanomaterials and products.
The following materials/products were included in the submitted dossier:
Examples of nano-sized ZnO raw materials (powder):
Z-COTE® (ZnO)
BASF SE
Nanox (ZnO)
Elementis
Nano TEC® 50 (ZnO)
Grillo Zinkoxid GmbH
Nano TEC® 60 (ZnO)
Grillo Zinkoxid GmbH
FINEX-50 (ZnO)
Sakai Chemical
MZ 30 (ZnO)
Tayca
Zinc Oxide Neutral (ZnO)
Symrise GmbH
Zano® 10 (former Zano®, ZnO)
Umicore
Z-COTE® HP1 (ZnO coated with triethoxycaprylylsilane)
BASF SE
Z-COTE® MAX (ZnO coated with dimethoxydiphenylsilanetriethoxycaprylylsilanecross-polymer)
BASF SE
Zinc Oxide NDM (ZnO coated with dimethicone)
Symrise GmbH
Zano® 10 Plus (former Zano® Plus, ZnO coated with octyl triethoxy silane)
Umicore
Examples of final cosmetic products with nano-sized ZnO raw materials:
W/O emulsion, uncoated ZnO (Sun Defense SPF 30/PA++, Ponds, 8.4% ZnO)
Unilever
W/O emulsion, coated ZnO (20% ZnO)
Umicore
O/W emulsion, coated ZnO (9% ZnO)
Umicore
O/W emulsion, coated ZnO (Perfect results SPF 15/PA++, Ponds, 2.2% (Z-COTE® HP)
Unilever
O/W emulsion, coated ZnO (all day moisture Lotion SPF 15, Olay, ZnO (Z-COTE® HP1)
Procter & Gamble
Comment
The data provided on the safety evaluation are mainly on uncoated ZnO. According to the information in submission II, the currently used coating materials are not UV absorbers and are common cosmetic ingredients that are widely used for different purposes in cosmetic products. The coatings consist of organic and inorganic materials such as silica, dimethicone, dimethoxydiphenylsilane and triethoxycaprylylsilane.
(References: 3, 4, 15, 16, 17, 18, 21, 24, 41, 51, 52, 84, 102, 103, 111, 112, 117)
CAS/EC number
| CAS No.: | 1314-13-2 |
| EC No.: | 215-222-5 |
Structural formula
Formula: ZnO
Crystallinity: Wurtzite
Empirical formula
Formula: ZnO
Physical form
Solid, white odorless powder.
Molecular weight
Molecular weight: 81.38 g/mol.
Purity, composition and substance codes
The purity has been reported to be ≥99.0% of zinc oxide for the various nanomaterials included in the dossier.
(References: 10, 11, 51, 54, 55, 108, 112)
Comment
Two other references (52, 109) indicated for evidence of purity do not provide information on the purity of the nanomaterials.
A number of tests have been performed with a nano zinc oxide (Finex-50) with an indicated purity of ≥96%. However, in the description of the physico-chemical characteristics of the ZnO materials information on the purity has not been provided.
Impurities/accompanying contaminants
No information has been provided on the presence of impurities/contaminants.
Comment
As a purity of ≥ 99% was demonstrated in the references below, the impurities/contaminants are ≤1%.
(References: 10, 11, 51, 54, 55, 108, 112).
Two other references (52, 109) indicated do not provide information on the purity of the nanomaterials.
Solubility
Solubility was determined by mixing 10 grams of zinc oxide in 100 ml of demineralized water for 30 minutes at room temperature. After mixing, the suspension was centrifuged for 6 hours to separate the (undissolved) zinc oxide particles from the solution. After centrifugation the solutions were not completely clear optically, and a blue haze was noticeable. A sample of the solution was used to analyze the Zn content using Inductively Coupled Plasma (ICP) spectroscopy. Only uncoated preparations of zinc oxide were analyzed. The possible effect of coating on solubility was not determined.
The zinc concentration in the aqueous solution was:
| Zinc oxide A: | Zano 10 (uncoated, Umicore), batch: Z08009 | 42 mg/L |
| Zinc oxide B: | NanoTec 50 (uncoated, Grillo), batch: MZ6362 | 20 mg/L |
| Zinc oxide C: | Z-COTE (uncoated, BASF), batch: EHHG1407 | 47 mg/L |
| Zinc oxide D: | FINEX-50 (uncoated, Sakai Chemicals, Japan), batch: 9Z22 | 27 mg/L |
(Reference: 115)
The solubility of pigmentary, coated and uncoated ZnO was also determined in water and tissue culture medium. In these assays, atom absorption spectroscopy was used for the determination of the Zn content.
The following results were obtained:
| Sample | Medium | pH | Solubility based on Zn concentration |
| ZnO (pigmentary, Fluka) | Aqua dest. | 6.5–7 | <0.1 mg/L |
| ZnO (pigmentary, Fluka) | DMEM/FCS** | About 7.4 | 35 mg/L |
| ZnO (uncoated, Z-COTE, BASF) | Aqua dest. | 6.5–7 | 1.5 mg/L |
| ZnO (uncoated, Z-COTE, BASF) | M4 medium* | About 7.4 | 1.2 mg/L |
| ZnO (uncoated, Z-COTE, BASF | DMEM/FCS | About 7.4 | 37 mg/L |
| ZnO (coated, Z-COTE HP1, BASF) M4 medium | About 7.4 | 2.4 mg/L | |
* M4 medium: reconstituted water containing salts and trace elements (water hardness: 2.5 mmol/L; alkalinity: 0.9 mmol/L).
** DMEM/FCS: Dulbecco/Vogt modified Eagle’s minimal essential medium/fetal calf serum. (References: 8, 9)
At low pH simulating stomach environment at pH 2.7, the solubility of particles ranged from 89.6% (particles >3 mm) to 98.5% (particles <1 mm).
(Reference: 57)
Comment
For the tests with uncoated ZnO (Reference: 115) some haze remained visible after centrifugation indicating that some non-dissolved ZnO particles remained in dispersion. The visible haze is very likely to be due to agglomerates/aggregates since dispersed nanoparticles would not be visible. These were also measured by ICP spectrometry. Thus, there is an overestimation of the solubility as determined by the method used. The data provided strongly suggest that equilibrium solubility had been reached, although it remains unclear if the solution was saturated, or if the particles remained undissolved because of the presence of some surface layer.
The data provided suggest that:
- the solubility of the nano-sized formulation in water is about 10× that of the pigmentary grade,
- in tissue culture medium the solubility of nano-sized and pigmentary grade is similar.
- the solubility of the coated and uncoated Z-COTE ZnO shows only a twofold difference, the coated ZnO being slightly more soluble. It should be noted that for the solubility of Z-COTE, two different solubility values (1.5 mg/L and 47 mg/L) have been provided and no explanation of this difference is given.
Additional information was provided on the effect of particle size on solubility in a simulated stomach environment (pH 2.7). Particles with a size <1 mm showed 98.5% dissolution. As the information on size was limited to <1 mm, no relevant information was provided specifically on nano-sized ZnO. This high solubility at low pH of both nano- and micron-sized ZnO particles was also recently demonstrated by Li et al. (Reference: AR16). In view of this, the SCCS considers that the solubility of ZnO in a stomach environment can be assumed to be high.
In conclusion, the data provided on solubility are inadequate to draw conclusions on the dissolution of ZnO nanoparticles. It is the view of the SCCS that the solubility data as presented are insufficient. Furthermore, to enable proper estimation of consumer exposure to any partially dissolved and/or insoluble fractions, the information for nanomaterials to be used as cosmetic ingredients should be provided not only on solubility, but also on the dissolution rate since equilibrium solubility is normally not achieved in the body (see SCCS Guidance on the Safety Assessment of Nanomaterials in Cosmetics SCCS/1484/12).
However, some additional information on solubility of ZnO nanoparticles in general is available in the open literature. The literature indicates that solubility of ZnO nanoparticles is in the same range as reported in the dossier (References: AR15, AR16, AR17, AR19, AR21). Some of the data indicate that the solubility of uncoated and coated ZnO (Reference: AR15), or ZnO either as nanoparticles or micron- sized particles, is similar and pH dependent (References: AR16, AR17). Also the solvent itself (water, tissue culture medium, presence of citric acid) influences the dissolution of ZnO nanoparticles (Reference: AR15, AR19, AR21). Reed et al. (Reference: AR21) compared dissolution rates between nano- and bulk-sized ZnO, and showed that the nano-form has a higher dissolution rate.
In view of the available information on the solubility of ZnO in aqueous media, it can be expected that very small concentrations of ZnO (e.g. <50 mg/L) will be in the form of a completely dissolved solution under static equilibrium conditions. This means that ZnO in a formulation will be simultaneously present as a mixture of solubilized Zn ions and a major part as undissolved ZnO particles - both with different diffusion and uptake characteristics.
The dissolution rate, also applicable to nanoparticles, describes the solubilization of ZnO nanoparticles in a certain fluid matrix over time, and will differ greatly depending on the fluid composition. In particular, the dissolution rate will depend critically on the fractional saturation of the matrix with Zn ions and the potential mechanisms for diffusion or active transport of Zn and O (and other) ions typical for biological processes. The dynamic nature of dissolution rate of any solubilization process implies that any mass fraction of Zn ions removed or washed away will lead to further dissolution and eventually complete solubilization of the particulate fraction of ZnO. Thus, under non-static conditions, e.g. in biological environments, ZnO nanoparticles will keep on releasing a certain mass fraction of Zn ions until they are completely solubilized.
(References: 8, 9, 57, 115)
Partition coefficient (Log Pow)
Log Pow: According to the applicant, measurement of the partition coefficient is not applicable as ZnO is practically insoluble in water.
Comment
This statement cannot be accepted in view of the partial solubility.
Note: depending on the coating on the particles, a partitioning of particles between aqueous and octanol phases may also be expected.
Additional physical and chemical specifications
Melting point: 1,975°C
Boiling point: -
Flash point: -
Vapour pressure: -
Density: 5.47 g/cm2
Viscosity: -
pKa: -
Refractive index: -
Particle size
The particle size distribution was provided for four uncoated ZnO nanoparticles designated A, B, C, D:
Zinc oxide A: Zano 10 (uncoated, Umicore), batch: Z08009
Zinc oxide B: NanoTec 50 (uncoated, Grillo), batch: MZ6362
Zinc oxide C: Z-COTE (uncoated, BASF), batch: EHHG1407
Zinc oxide D: FINEX-50 (uncoated, Sakai Chemicals, Japan), batch: 9Z22
To determine particle size distribution, 30 w% zinc oxide dispersions were prepared by adding zinc oxide to a mixture of water and dispersing agent. The dispersion was composed of ZnO (150 g), Tego Dispers 752W1 (30 g) and water (320 g). The dispersion was premixed in a dissolver and transferred to Dispermat SL-12 bead mill thereafter. The bead mill was operated using 0.3 mm YTZ beads at a filling ratio of 85% and a rotor speed of 4,500 rpm. The zinc oxide suspensions were circulated through the mill over a period of 60 minutes. After bead milling, a sample of the suspension was collected for particle size analysis.
The particle size distribution was measured using a CPS Disc Centrifuge (model DC20000)2, operated at a constant rotation of 10.000 rpm. 0.1 g of the 30 w% suspension was diluted in 30 ml water, after which 0.15 ml of the dilution was injected into a rotating disc and particle size measurements were recorded. The results are expressed as weight average and number average particle size.
The applicant claims that the particle size distribution (PSD) determination in aqueous environments can only be done for uncoated ZnO and considers it not relevant to determine the PSD for coated ZnO. It is argued in the submission that the coating would not change the particle size of the ZnO particle. This is supported by electron microscopy images where no differences in particle size and morphology can be identified between coated and uncoated materials. The applicant recognized that in cosmetic formulations the particle size distribution of coated and uncoated ZnO may be of interest, but the methodology will only provide information very close to the primary particle size. It is nevertheless unlikely that normal coatings will bring any significant change to particle size distribution of a nanomaterial.
For all ZnO preparations, the number distribution results show that 90% of the particles have a size below 100 nm, whereas for the weight distribution the percentage of particles with a size below 100 nm varies (ZnO A d75, ZnO B d75, ZnO C d25, ZnO D d90).
The median particle size distribution (the D50 indicates that 50% of the particles have a size below that measurement) of the four evaluated ZnO nanomaterials ranged from 30 nm (ZnO D) to 55 nm (ZnO C).
Additional parameters of the ZnO nanomaterials such as number of particles per mass unit, and surface area per mass unit were not provided.
(Reference: 114)
Microscopy
The ZnO nanoparticle distribution of the formulations was investigated with two different electron microscopy techniques: Cryo-SEM and ambient TEM. Several examples of both EM and TEM evaluations are presented.
TEM evaluations of uncoated ZnO
A) Z-COTE®, uncoated ZnO (BASF, left image, TEM, Reference: 13)
ZnO particles have different shapes (rod-like and isometric) and a broad size distribution range of 30–200 nm. The ZnO particles were generally present as clusters.
B) Z-COTE® HP1, coated ZnO (BASF, right image, TEM, Reference: 13)
ZnO nanoparticles have different shapes (rod-like and isometric) and a broad size distribution range of 30–200 nm. The ZnO particles were generally present as clusters.
Comment
Both uncoated and coated Z-COTE® ZnO particles show similar morphology in the TEM images.
TEM evaluation of ZnO in formulations
A) W/O emulsion, coated ZnO (20% ZnO, Umicore, left image, TEM, Reference: 18)
ZnO nanoparticles have different shapes (rod-like, star-like and isometric) and a broad size distribution range of 30–150 nm. The ZnO particles were present mainly as clusters.
B) O/W emulsion, coated ZnO (9% ZnO, Umicore, right image, TEM, Reference: 25)
ZnO nanoparticles have different shapes (rod-like, star-like and isometric) and a broad size distribution range of 30–150 nm. The ZnO particles were present mainly as clusters. In addition, spherical particles with a size between 200 nm and 1 μm were present.
Comment
The TEM image of O/W emulsion (right image, above) as presented in the dossier could not be found in the cited reference (Reference: 24) but it is contained in Reference: 25. There is a similar discrepancy in Reference: 24 which refers to SEM images, whereas the image presented above shows TEM results.
The various formulations presented all showed the presence of ZnO particulates mainly as clusters, with size ranges as indicated for the raw materials. These clusters may be present in the original formulation, but it is also possible that it is due to the excessive load of the EM sample. Z-COTE® and Z-COTE®HP1 with a size of 30–200 nm show clusters with rod-like, star-like and isometric forms. Umicore ZnO with a size of 30–150 nm also shows clusters with rod-like, star-like and isometric forms. Similar results are shown for both O/W and W/O emulsions.
The star-like forms present in the formulations are not observed in the raw material for Z-COTE®. Both ZnO nanomaterials contain rod-shaped particles for which the Z-COTE® ZnO nanoparticles show more irregular shapes compared to the Umicore ZnO nanomaterial.
(References: 13, 18, 25)
SEM evaluation of uncoated ZnO nanomaterials
Comment
The SEM images of the three examples show similar morphology of the uncoated Nano Tec® 50 and Zano® 10 nanomaterials.
SEM images of ZnO nanomaterial in various formulations were included in the dossier submitted by the applicant. As these are relevant for final cosmetic products they have been considered in this opinion as additional background information but have not been further evaluated.
(References: 58, 109, 110)
Crystal structures
X-ray diffraction (XRD) analysis was used for determination of the crystal structure. Rietveld refinement characterization was used for analysis of crystallite size.
The crystal structure for all samples was identified as purely hexagonal zinc oxide. For zinc oxide this is often referred to as a "Wurtzite” structure (see also section 3.1.1.5). The crystallite sizes determined via Rietveld refinement characterization were as follows:
| Zinc oxide A: | Zano 10 (Umicore), batch: Z08009 | 48 nm |
| Zinc oxide B: | NanoTec 50 (Grillo), batch: MZ6362 | 36 nm |
| Zinc oxide C: | Z-COTE (BASF), batch: EHHG1407 | 67 nm |
| Zinc oxide D: | FINEX-50 (Sakai Chemicals, Japan), batch: 9Z22 | 17 nm |
(References: 53, 116)
UV absorption
UV absorption was determined for four uncoated ZnO nanomaterials. A suspension of zinc oxide samples was prepared using a mild bead mill operation to break up agglomerates and stabilize the individual particles as much as possible. These dispersions were then used to measure the UV/vis absorbance and reflection spectra. The 30 w% dispersions were diluted to a concentration of 0.1 g/L ZnO and the UV/vis absorption spectrum was measured using a Genesys 6 UV/vis spectrometer.
The following ZnO preparations were analysed:
Zinc oxide A: Zano 10 (uncoated, Umicore), batch: Z08009
Zinc oxide B: NanoTec 50 (uncoated, Grillo), batch: MZ6362
Zinc oxide C: Z-COTE (uncoated, BASF), batch: EHHG1407
Zinc oxide D: FINEX-50 (uncoated, Sakai Chemicals, Japan), batch: 9Z22
Comment
A similar pattern was observed regarding the UV absorption for the four evaluated ZnO nanomaterials. However, only uncoated ZnO materials were analysed.
Zeta potential
Zeta potential was determined using electrophoresis (Zetasizer Nano from Malvern). The pH was adjusted with NaOH.
The following samples were investigated:
FINEX-50 (Sakai Chemicals, Japan, supplied by Umicore)
Zano 10 (Umicore)
NanoTec 50 (Grillo)
Z-COTE (BASF, batch: EHH/0201)
There was no significant difference between the samples in terms of the charge.
The curve shapes are characteristic for ZnO powders. The iso electric point (IEP) was in the range of pH 9.5–10.
(Reference: 31)
Droplet size in formulations
The applicant provided additional information on the effects of various formulation and technical parameters (sunscreen type, nanomaterial concentration, viscosity, propellant gas, actuator and valve type) on size and size distribution of droplets generated when a sunscreen is used as an emulsion for spray application. The information was provided as the results of an investigative study (Reference: 38).
Different types of emulsion; water in silicone (W/Si) and water in oil (W/O), were prepared using high concentrations of organic and inorganic UV-filters (ZnO as Z- COTE MAX, 1% in W/Si emulsion, 1% in W/O emulsion, among others). These formulations were incorporated in aerosol cans with gas (a blend of butane, propane and isobutane).
The size and size distribution of the droplets were analyzed by laser diffraction using a Malvern® Spraytec.
The measuring distance of the droplets was set at 11 cm for all the experiments. In general, assessments for these types of evaluations and measurements are carried out at a distance of 30 cm from the laser beam. However, the authors of the paper considered that 11 cm was a more common distance for normal use of sunscreen sprays.
The results showed that the sprayability of the formulation and the particle size characteristics of the emitted sprays are dependent on the physicochemical properties of the formulations. Sprayable waterproof sunscreen emulsions with a high sun protection factor and a low (<5%) percentage of emitted droplets below 30 μm could be developed by optimizing formulation parameters and using appropriate actuators and valves. For all experiments, the percentage of droplets below 10 μm was less than 0.1%. Droplet size and size distribution of the emitted droplets were highly influenced by all the parameters studied and especially by propellant concentration, viscosity, presence or absence of inorganic filters (powder) and the design of valves and actuators. Considering the aspect of the final product and droplet size, the best products (in terms of optimal droplet size) were obtained with the lower gas concentration (22%), higher concentrations of inorganic sunscreen filter and higher viscosities. The final choice of the valve and actuator was made based on the aspect of the final product after spraying. Therefore, when sunscreen-containing aerosols are formulated it is important to consider each technical and formulation parameter in order to reach the optimum size and size distribution of droplets.
(Reference: 38)
According to the applicant, no sunscreen spray products containing nano-sized ZnO are available on the EU market. It was stated that if such sprayable sunscreen products were to be produced and marketed they would comply with current standards and requirements according to the relevant guidance documents as referred to below.
- The Technical Guidance Document on Risk Assessment of the European Chemical Council emphasized that aerosols with a Mass Medium Aerodynamic Diameter (MMAD) greater than 10–15 μm are not respirable for humans, i.e. the aerosol particle sizes above this cut-off will deposit in the upper regions of the lungs due to their large particle sizes. Only particles with a size less than 10–15 μm reach the gas-exchange region of the lung.
- The U.S. Silicones Environmental, Health and Safety Council (SEHSC) recommends that, "When considering a consumer aerosol application for any silicone-based material, regardless of the method of aerosol generation, the particle size MMAD should be at least 30 μm with no more than 1% of the particles having an aerodynamic diameter of 10 μm or less. Following this guidance ensures that virtually all aerosol particles will be trapped in the nasopharyngeal region and very few if any particles will be deposited in the tracheobronchial region.”
(References: 43, 45, 86)
Comment
The results indicate that mainly larger droplets were present when spraying a water- or silicone-based nanoparticle containing test aerosol at a distance of 11 cm. The small fraction of droplets with a diameter less than 30 μm was based on droplet size distribution measured by weight. It should be noted that even a low fraction based on aerosol weight is relevant as it contains a high number of droplets. At larger distances such as 30 cm which is normally used for such investigations, a short time lapse may occur allowing for evaporation of aerosol liquid components resulting in smaller droplet sizes than measured at 11 cm. These dried residual particles spreading in the air also need to be taken into account for possible inhalation exposure. Notably, this mechanism is even more relevant when high vapour pressure fluids such as alcohols are used in the generation of the aerosol, compared to when water or oil are used as dispersion fluids.
The report indicates that in certain tests, an inorganic filler such as TiO2 or ZnO (Z-COTE max) was used. However, it was not indicated in the results section which inorganic filler was used in the experiments for which the data were presented. Therefore, the data cannot be exclusively ascribed to ZnO nanomaterial.
In view of the presented data, the possibility that the dried residual aerosol particles may lead to lung exposure after inhalation needs to be taken into account. Therefore the measurement of the droplet size distribution needs to be complemented by measurements of the size distribution of the dried residual aerosol particles as well.
Homogeneity and stability
Photostability
Method
Photostability was measured by UV absorption according to a BASF internal method: PMMA plates (Helioscreen HD6, 6 μ m), 1.3 mg/cm2 application rate, four plates and 11 scans/plate, measurements with Labsphere 1000S.
Samples
O/W emulsions with uncoated and coated ZnO
| Formulation A: | Z-COTE (10%, uncoated, BASF): | UV10037-1-1 (SPF 6.6) |
| Formulation B: | Z-COTE HP1 (10%, coated, BASF): | UV 10037-2-2 (SPF 6.6) |
| Formulation C: | Zano 10 Plus (20%, coated, Umicore): UM08-ZP20-OW (SPF 31.3) | |
Light source
Atlas CPS+ with Atlas filter solar standard according to DIN 67501.
The O/W formulations were photostable over the whole UV range (from 290 to 400 nm).
However, according to the applicant, few absorbance curves were higher following irradiation as it was technically not possible to measure exactly the same point on a PMMA plate before and after irradiation, and also in case the formulation spreading of the plate was not homogenous. This is known to result in slightly different absorbance behaviour. The observed minimal differences could be considered as an indication of variation in the measurement technique and not as variation in the photostability of the nanomaterials.
It was concluded by the applicant that all three model O/W formulations with uncoated (Z-COTE) and coated (Z-COTE HP1, Zano 10 Plus) ZnO were photostable over the whole UV range between 290 and 400 nm.
(Reference: 29)
Summary on supplementary physicochemical characterization
The comparative analysis and presentation of the physicochemical characteristics of the representative selected nano-sized ZnO materials shows that all nano-sized ZnO materials as described in the dossier can be considered to be broadly similar, irrespective of the manufacturing processes used (wet chemical or pyrolytic). The crystal structure of all of the ZnO nanomaterials included in the dossier is the same (wurtzite/hexagonal) and the particle shapes are roughly similar. The particle size distributions for all of the products fall within the same range and so do all other parameters tested such as solubility, zeta potential and photostability. According to the applicant, the set of physicochemical data on representative nano-sized ZnO raw materials, as well as on examples of additional nano-sized ZnO raw materials found on the EU market, indicates that the nano-sized ZnO products can be considered to be similar overall.
Comment
It should be noted that it is technically possible to produce nano-dispersed ZnO with different physicochemical properties to those described in this dossier. Therefore, this opinion specifically relates to those coated and uncoated ZnO nanomaterials for which data have been submitted in this dossier. It is required that manufacturers carefully analyse the raw materials which they obtain, particularly if they come in a finally stabilized formulation in fluids which may contain different forms/surface modifications of the nanomaterials. Thus, this assessment applies to ZnO nanoparticles similar to those included in this dossier, with the following characteristics:
- ZnO nanoparticles of purity ≥99%, with wurtzite crystalline structure and physical appearance as described in the dossier, i.e. clusters that are rod-like, star-like and/or isometric shapes.
- Median diameter (D50: 50% of the number below this diameter) of the particle number size distribution is between 30 nm and 55 nm, whereas the D1 (1% below this size) is above 20 nm.
- ZnO nanoparticles that are either uncoated or coated (with triethoxycaprylylsilane, dimethicone, dimethoxydiphenylsilanetriethoxycaprylylsilane cross-polymer, or octyl triethoxy silane).
- ZnO nanoparticles that have a comparable solubility to that reported in the dossier, i.e. below 50 mg/L (approximately the maximum solubility of the ZnO nanomaterials for which data are provided in the dossier).
Regarding the characterization of the ZnO nanomaterials, product parameters such as the number of particles per mass unit and surface area per mass unit were not provided. It is at the moment under consideration that mass (weight) alone may not be the best parameter for the description of a dose response relationship in toxicity studies. Therefore, it is the view of the SCCS that additional parameters, such as number of particles per mass and surface area per mass, are usefull additions to the characterization of the nanomaterials. Where such parameters are available, it is possible to evaluate dose response relationships for toxicity by these alternative dose parameters (number of particles, total surface area administered) as well.
La estructura de tres niveles utilizada para comunicar la opinión del Comité Científico de Seguridad de los consumidores (SCCS) es propiedad intelectual de Cogeneris SPRL.