Filter by
JRC news and updates (1789)
RSS
Novel therapy used in an ongoing Phase I clinical study shows substantial antitumour effect in patients with metastatic castration-resistant prostate cancer (mCRPC).

The Global Report on Food Crises identifies conflicts as major driver, and the Gaza Strip food crisis as the worst in eight years.

A harmonised approach is a first step in eventually establishing exposure levels in EU tap water.

First 10 manufacturers commit to develop interoperable connected products within a year.

Warm spring weather benefitted crops in most of Europe

Favourable winter crops outlook in Türkiye
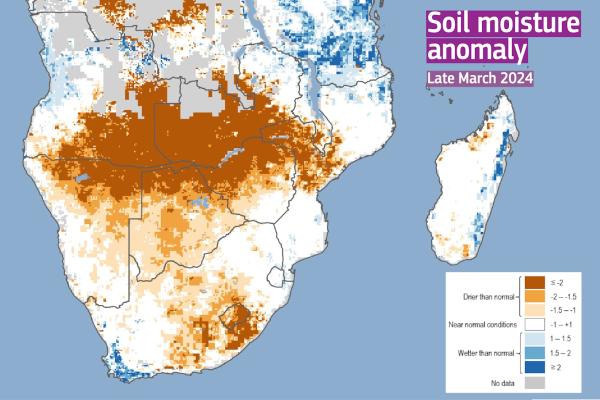
Zimbabwe, Zambia, and Malawi have officially declared state of emergency.
What are the most promising current and emerging technologies when the next airborne disease hits?

There is a pressing need to shed light on net-zero emissions pledges by cities. JRC scientists propose a framework to establish qualitatively the level of unavoidability of residual emissions.

The report provides examples of how the JRC has made a positive impact on policymaking and society in the past year.
